Diatomic components are molecules composed of only two atoms, of the same or different chemical elements. If a diatomic molecule consists of two atoms of the same component, like hydrogen (H2) or oxygen (O2), then it’s said to be homonuclear. Otherwise, in case a diatomic molecule consists of two different atoms, such as carbon dioxide (CO) or nitric oxide (NO), the molecule is supposed to be heteronuclear.
Diatomic Elements include two atoms bonded together. In contrast, monatomic elements include single atoms. Many substances are diatomic, such as HCl, NaCl, and KBr. Diatomic compounds consist of two distinct elements. You will find seven pure components that form diatomic Elements.
Diatomic Elements Definition
There are major segments that form the planet’s air: oxygen, nitrogen, and even a tad sample of argon. To be better precise, nitrogen constitutes 78% of the space whilst oxygen makes up 21%. This oxygen, O2, and nitrogen, N2, have something in common: Both are known as diatomic elements.
As pure components, diatomic elements are just seven specific molecules that are composed of two atoms. Each one of the elements in this noble group is gases. If we notice the prefixed from the word diatomic, it comes from the Greek source of two.’ One big way to understand you are functioning with a diatomic element is to think about its own formulation.
Every molecule has its own unique molecular formula, also for diatomic components, their formula regularly contains an addendum of 2, which performs two atoms in its arrangement. For example, the diatomic component oxygen has a formulation of O2, meaning that there are two different oxygen atoms present.
At room temperature, you will find five diatomic elements, all of which exist at the gas type: hydrogen, nitrogen, oxygen, fluorine, and chlorine. At room temperature, bromine and iodine commonly exist in the liquid kind. However, with this greater temperature, they will also exist as gases.
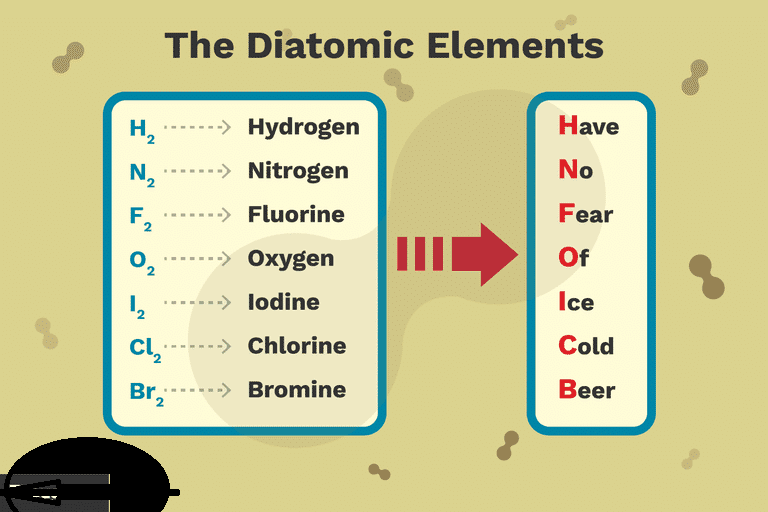
Diatomic elements are unique since the atoms which form them do not like to be alone. That is, you’ll never find a nitrogen or fluorine atom, for instance, hanging out. Instead, these molecules will always be paired together since they need to pool resources to possess enough electrons. 1 great way to remember that atoms form the blessed seven diatomic elements is to think of another mnemonic device: ”I Bring Cookies For Your New Home”.
If there’s ever a need to recognize these diatomic elements on the periodic table, they can be spotted by remembering the “seven rule” as well. Shown in the case, should you take your finger and draw the number seven, beginning at nitrogen, you will see that six of the diatomic elements are arranged in the form of a number seven. When drawing the number seven don’t neglect the seventh element, hydrogen.
What Are The 7 Diatomic Elements?
This is a list of those seven diatomic elements. The seven diatomic components are:
Hydrogen (H2)
Nitrogen (N2)
Oxygen (O2)
Fluorine (F2)
Chlorine (Cl2)
Iodine (I2)
Bromine (Br2)
All these elements are nonmetals because halogens are a special kind of nonmetallic element. Bromine is a liquid at room temperature, whereas the other components all gases under ordinary conditions. As the temperature is lowered or pressure is increased, the other components become diatomic liquids.
Astatine (atomic number 85, emblem At) and Tennessee (atomic number 117, symbol Ts) will also be from the halogen group and might form diatomic molecules. However, some scientists forecast Tennessine may behave much more like a noble gas.
Whilst just these seven components regularly form diatomic molecules, other elements can form them. However, diatomic molecules shaped by other elements aren’t very secure, so their bonds are easily broken.
How To Remember?
There’s an effortless method to recall the seven diatomics, the “seven principles ” To begin with, visit element seven (Nitrogen) and then make a seven with your finger through oxygen and fluorine, down through chlorine, bromine, and iodine. That is six. The hydrogen is that the”oddball” of the table, off alone. Suggested To Read –What Is A Hypotonic Solution
Which Are Diatomic Elements?
At Room, TemperatureThere Are Five Diatomic Elements, All Of Which Exist In The Gas Form: Hydrogen, Nitrogen, Oxygen, Fluorine, And Chlorine.
Are Diatomic Compounds?
All Compounds Are Molecules, But Not All Of the Molecules Are Compounds (Should They Happen To Have Atoms Of The Exact Same Element). Diatomic Molecules Are Molecules Composed Of Just Two Atoms Of The Exact Same Or Different Elements. However, Not Each Diatomic Molecule Comprises A Chemical Compound.
Just how Many Diatomic Elements Are There?
There are just seven diatomic elements present out there in the world.
Can Be Hcl A Diatomic Molecule?
All Other Diatomic Molecules Are Chemical Compounds Of Two Different Elements. Many Elements Can Combine To Form Heteronuclear Diatomic Molecules, Based On Temperature And Stress. Related – How to Balance Chemical Equations